With $600M valuation, Elpiscience seeks HKEX listing as pioneer MCE-focused biotech
On November 24, Elpiscience Biopharmaceuticals filed a new application for a main board listing on the HKEX, with CITIC Securities acting as the sole sponsor.

Founded in 2017, Elpiscience Biopharmaceuticals is dedicated to developing next-generation immuno-oncology therapies. The company focuses on converting "cold tumors" (characterized by poor T-cell infiltration and limited response to PD-1/PD-L1 inhibitors) into "hot tumors" (rich in effector T cells and responsive to immunotherapy), addressing significant unmet clinical needs.
With its differentiated R&D strategy and technological potential, Elpiscience has attracted sustained interest and investment from leading domestic and international institutions. These include Lilly Asia Ventures (LAV), Hillhouse Group, Tencent, CGTI Fund, and Hyfinity Investments. According to its latest prospectus, the company has completed four financing rounds, raising approximately USD 252 million. Its post-money valuation after the most recent round reached USD 600 million. Regarding shareholding structure, LAV USD, an investment platform under Lilly Asia Ventures, holds 22.93% and is the largest single shareholder.
Three Major Antibody Platforms Drive Pipeline Innovation
Elpiscience Biopharmaceuticals' product portfolio is not a simple collection of disparate assets, but is built upon an integrated R&D engine that spans "target discovery — molecular design — translational medicine — clinical development." Supported by this core platform, Elpiscience has established a differentiated pipeline centered on immunomodulation, covering two major therapeutic areas: oncology and autoimmune diseases. The current pipeline comprises six key assets, four of which are in clinical stages and two in preclinical development.
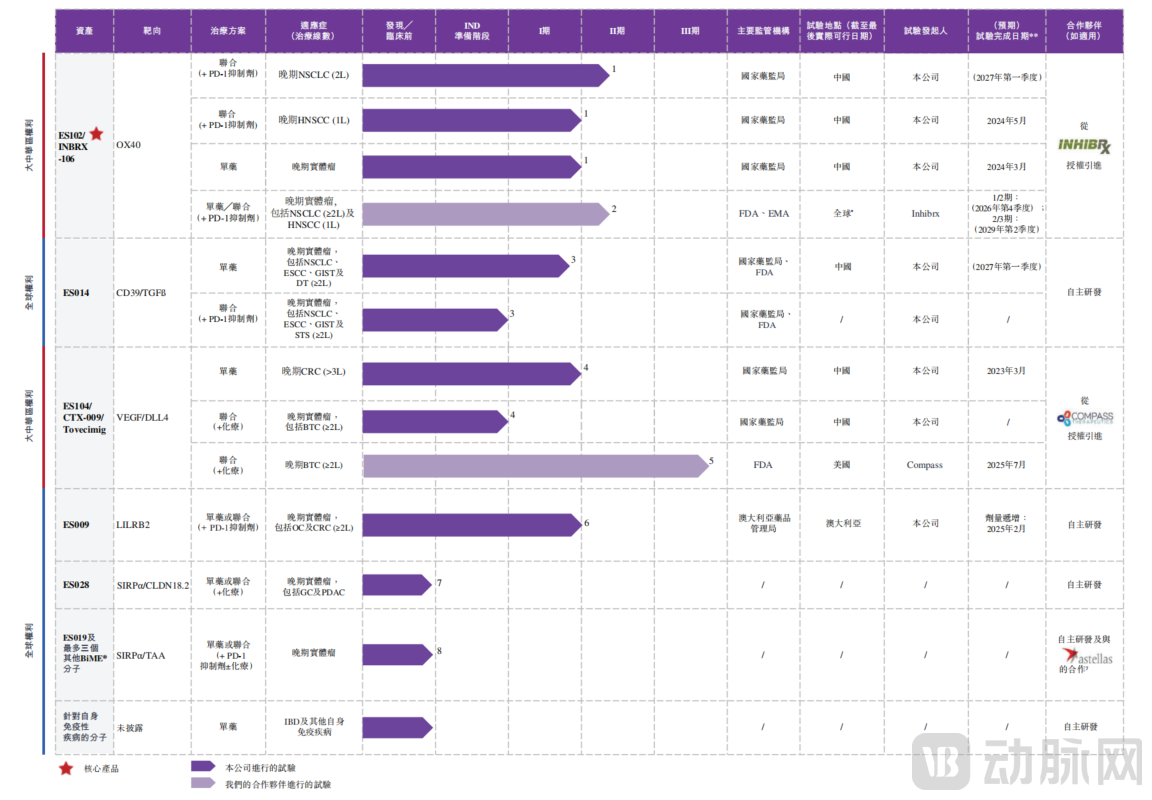
Elpiscience's R&D Pipeline Status
As a core asset, ES102 is one of the few hexavalent OX40 agonists globally that has entered Phase II or later clinical stages. Its development focuses on improving therapeutic outcomes for patients with poor response to immune checkpoint inhibitors.
OX40 serves as a key co-stimulatory receptor for enhancing adaptive immune responses. Traditional agonists have long faced challenges in balancing activation strength, durability, and toxicity. Leveraging the company's engineering platform, ES102 achieves mechanistic innovation through its hexavalent binding design that enhances signal transduction. This approach both strengthens the proliferation and killing capacity of effector T cells and reverses immunosuppression mediated by regulatory T cells, with the dual mechanisms synergistically breaking through industry bottlenecks.
In clinical development, the efficacy potential of combination therapies is becoming evident: In China, two Phase I studies of ES102 monotherapy and combination therapy with a PD-1 antibody have been completed, and a Phase II trial combining ES102 with toripalimab for advanced non-small cell lung cancer was initiated in April 2025. In the United States, partner Inhibrx has advanced to Phase I/II, initiating a Phase II/III registrational trial in 2024 combining ES102 with Keytruda as first-line treatment for head and neck squamous cell carcinoma.
Other clinical-stage assets each possess differentiated advantages: ES014 is the first-in-class CD39/transforming growth factor beta (TGF-β) bispecific antibody to enter clinical studies globally; ES104 is a differentiated bispecific antibody and one of only two vascular endothelial growth factor (VEGF)/delta-like ligand 4 (DLL4) bispecific antibodies in active clinical development worldwide; ES009 is a differentiated monoclonal antibody (mAb) targeting leukocyte immunoglobulin-like receptor B2 (LILRB2) with best-in-class potential.
In preclinical research, Elpiscience leverages systematic early mechanistic research approaches and integrated technology platforms to advance multiple promising drug candidates including ES028, covering novel target treatments for gastric cancer, pancreatic ductal adenocarcinoma, non-small cell lung cancer, head and neck squamous cell carcinoma, hepatocellular carcinoma, colorectal cancer, and other cancer types.
The continuous output of this differentiated pipeline fundamentally relies on the company's systematic platform capability centered on MCE (Myeloid Cell Engager). While novel immune cell engagers have become the next generation of hot therapeutics, most companies focus on TCE (T Cell Engager) primarily for hematological tumors, facing challenges in solid tumor breakthroughs. Elpiscience has taken an alternative path by developing the MCE field, becoming a rare global player.
As the core carrier of MCE technology, the BiME platform (Bispecific Macrophage Engager) specializes in reprogramming tumor-associated macrophages (TAM) through dual-targeting activation of macrophage function, addressing both anti-tumor effects and pathological B-cell clearance in autoimmune diseases, with safety and activity superior to traditional T-cell therapies. Combined with the Acebody platform's efficient production capability for IgG-like bispecific antibodies (bsAb), ElpiSource antibody library's precise target screening ability, along with translational medicine for accurate matching and efficient dual-track clinical advancement in China and the United States, these elements collectively form Elpiscience's unique technological barrier in the myeloid cell regulation field.
In global collaboration, Elpiscience has built a diversified partnership network through licensing and co-development. Through license agreements with Inhibrx and Compass, the company rapidly obtained development and commercialization rights for potential assets including ES102 and ES104 in Greater China, shortening R&D cycles. The combination therapy research with Junshi Biosciences focuses on the synergistic efficacy of ES102 and toripalimab, expanding clinical application scenarios. The collaboration with Astellas around the BiME platform and the joint establishment of the ELPITEX platform with Partex AI not only achieves complementary technical resources but also promotes breakthroughs in innovative target and antibody design technologies, laying the foundation for continuous pipeline iteration and long-term development.
Financially, as a clinical-stage company that has not yet achieved product commercialization, Elpiscience demonstrates typical R&D-driven operational characteristics. According to the prospectus, the company reported net losses of RMB 853 million and RMB 88 million in 2023 and 2024 respectively, with the core loss source being continuous R&D investment and related operational expenses. The company indicated in its prospectus that the net loss for 2025 is expected to expand compared to 2024, mainly due to two factors: first, the company recognized RMB 107 million in non-recurring income during the first nine months of 2024 (originating from the collaboration, option, and license agreement with Astellas), while no such income exists during the same period in 2025; second, as clinical and preclinical studies of pipeline drugs continue to advance, R&D costs will further increase.
Global Immunotherapy Enters the Multi-Target Era
The global immuno-oncology market continues to experience rapid growth, with immune checkpoint inhibitors represented by PD-1 antibodies becoming core growth drivers for major pharmaceutical companies. Taking Merck as an example, its PD-1 antibody Keytruda (pembrolizumab) achieved USD 29.5 billion in sales in 2024, reflecting the substantial global clinical demand and commercial potential of immunotherapy. Beyond established targets like PD-1/PD-L1, multinational pharmaceutical companies such as BMS, Merck, and Roche are actively advancing research on emerging immune checkpoints including LAG-3, TIM-3, and TIGIT. Simultaneously, strategies for multi-target modulation of the tumor microenvironment are increasingly becoming an industry consensus, with targets including CD39, TGF-β, LILRB2, and OX40 all entering accelerated development phases.
Regarding market size, according to QVIA data, global oncology drug spending reached USD 252 billion in 2024, with North America, Europe, and key Asia-Pacific countries accounting for over 70% of this total. In the Chinese market, continuously rising cancer incidence drives essential clinical demand, coupled with policy support and capital investment, accelerating the rise of the local innovative drug industry and creating broad development space for biotech companies like Elpiscience that focus on differentiated fields. In recent years, benchmark cases such as BeOne Medicines (formerly BeiGene)'s out-licensing of core assets to Novartis and Hengrui Pharma's multi-dimensional collaboration with Eli Lilly fully demonstrate that high-potential innovative pipelines with clinical value and technological barriers are consistently becoming the focus of global capital and pharmaceutical companies.
Against this industry evolution backdrop, Elpiscience's strategic path demonstrates strong foresight and consistency. The company focuses on the immune activation strategy of converting "cold tumors" into "hot tumors," building a differentiated pipeline around areas such as myeloid cell regulation (e.g., LILRB2), angiogenesis pathway remodeling (VEGF/DLL4 bispecific antibody), and immunosuppressive factor blockade (CD39/TGF-β bispecific antibody). This aligns closely with the current global trend in immunotherapy shifting from "single-target activation" towards "multi-pathway synergistic regulation." Its core products, including ES102, ES014, and ES104, have all entered the clinical validation stage, with some projects already receiving FDA IND approval.
Looking ahead, as more products advance into clinical trials, Elpiscience is expected to successively obtain key clinical readouts and gradually approach commercial inflection points. As an important representative of Chinese innovative enterprises in the global immuno-oncology field, whether Elpiscience can leverage its IPO to gain stronger resource allocation capabilities and achieve the leap from technological breakthrough to commercial realization warrants continued attention from both the industry and capital markets.
