Clinical breakthroughs and policy green lights: is China's BCI industry at a commercial turning point?
The year 2025 is universally regarded by the industry as the inaugural milestone for China's brain-computer interface (BCI) sector.
This consensus is solidly grounded. On the policy front, a series of pivotal measures have been implemented: In March, the National Healthcare Security Administration issued the Guidelines for Establishing Pricing of Medical Service Items in Neurology (Trial), marking the first inclusion of BCI-related procedures in national health insurance and establishing the foundation for a sustainable commercial model. In July, seven ministries, including the Ministry of Industry and Information Technology of the People's Republic of China and the National Development and Reform Commission, jointly released the Implementation Opinions on Promoting the Innovative Development of the Brain-Computer Interface Industry, formally elevating BCI to the status of a national strategic priority. Subsequently, in November, BCI was designated as one of the "Six Future Industries" in the framework of the China's 15th Five-Year Plan, underscoring its role as a new driver for economic growth.
In terms of capital investment, according to incomplete statistics from VCBeat, by November 20, 2025, the Chinese BCI sector had completed 28 financing rounds within the year, with total funds raised exceeding RMB 5 billion. This includes a record-setting RMB 350 million Series B financing round completed by StairMed Technology in February, which stands as the largest single financing round in China's BCI field to date.
Most critically, the clinical arena witnessed remarkable breakthroughs throughout 2025. In March, StairMed Technology successfully conducted China's first prospective First-in-Man (FIM) clinical trial for long-term implantation of an invasive BCI system at Huashan Hospital Affiliated to Fudan University. In September, a team led by Professor Duan Feng of Nankai University accomplished the world's first clinical trial using an interventional BCI to assist in motor function recovery of a paralyzed limb in a patient with hemiplegia. Also in September, Mindtrix Technology completed the world's first Investigator-Initiated Trial (IIT) for visual reconstruction capable of interpreting "complex graphics and multiple colors," achieving a leap from simple light perception to the recognition of intricate shapes and even color perception. In November, Neuracom Technology, in collaboration with Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, performed the first clinical implantation of a fully China-developed BCI chip, successfully capturing neural signals from the patient's sensorimotor cortex.
It is evident that 2025 holds profound historical significance for the BCI field in China. Many industry professionals share this view. Liu Bing, Founder of Mindtrix Technology, remarked, "This year has indeed seen numerous significant developments, collectively driving the BCI field to a critical inflection point—it is transitioning from an initial focus on exploring cutting-edge technologies to a new phase centered on demonstrating clinical value."
How, then, should this transition be specifically understood?
BCI After Five Years: Where Does It Deliver?
In January 2020, a collaborative team from Zhejiang University and the Department of Neurosurgery at the Second Affiliated Hospital Zhejiang University School of Medicine completed China's first clinical translational research project involving an implanted Brain-Computer Interface (BCI). This groundbreaking study enabled a patient with complete quadriplegia to control an external robotic arm and hand—performing tasks such as shaking hands, drinking a beverage, eating fried dough sticks, and playing mahjong—through "thought" alone, achieving three-dimensional movement control. This milestone is widely regarded as the genesis of China's BCI industry, providing the public with a tangible concept of the technology's potential.
In the subsequent years, BCI rapidly gained momentum in China, remaining a topic of intense interest. Media coverage primarily focused on technological and material innovations, speculative future clinical applications, and financing activities within the capital markets. However, over time, the industry's focus has progressively shifted toward clinical translation, raising a pivotal and increasingly prominent question: What are the definitive application scenarios for BCI, and what is their current status? The answer may lie in the series of breakthrough clinical studies witnessed this year.
BCI systems are categorized into three types based on the placement and method of electrode interaction with the cerebral cortex: invasive, non-invasive, and semi-invasive. Currently, non-invasive BCI dominates clinical applications, accounting for approximately 86% of the market. As these systems require only scalp-attached electrodes, they offer high safety and lower costs. Present primary applications include attention and cognitive training, closed-loop regulation of sleep and mood, and closed-loop neurorehabilitation. The latter integrates eye-tracking, electroencephalography (EEG), electromyography (EMG), and assistive rehabilitation devices for motor assistance or speech output.
Based on current market feedback, BCI applications in these areas are relatively mature. A key example is the first mass-produced intuitively controlled smart bionic hand launched by BrainCo. Users can perform fine motor skills like grasping, pinching, and rotating with the prosthesis upon wearing it, achieving an experience close to that of a natural limb. The company reports that quarterly sales of the smart bionic hand in 2024 have already surpassed the total sales for the entire year of 2023 by four to five times, indicating rapid market expansion.
Another representative non-invasive product category comprises various smart EEG headbands. For instance, the BrainLink device from Macrotellect utilizes a forehead electrode to capture EEG signals. When combined with multimodal data such as EMG and Heart Rate Variability (HRV), it enables functions like attention training, meditation assistance, and emotion regulation. Consequently, it is widely adopted in consumer-grade scenarios encompassing education, psychological intervention, and daily health management. In 2024, BrainLink sales exceeded 100,000 units, ranking among the top three in China's consumer-grade non-invasive BCI market.
In contrast, invasive and semi-invasive BCIs collectively hold a market share of only about 14%. However, they target clinical scenarios with profound, unmet needs. These include restoring motor function in patients with high-level paralysis, enabling high-speed "thought-to-text" communication for individuals unable to speak, visual function reconstruction for the blind, and exploring neuromodulation therapies for psychiatric disorders like depression and obsessive-compulsive disorder. The successful realization of any one of these applications would establish an entirely new medical industry sector.
Focusing specifically on invasive BCI, the direct implantation of electrodes into the brain cortex provides exceptionally high signal quality. However, this approach also introduces significant safety concerns, posing substantial technical challenges that have consistently constrained clinical research. Despite these hurdles, the seemingly futuristic clinical scenarios targeted by invasive BCI are gradually approaching reality, as evidenced by developments this year.
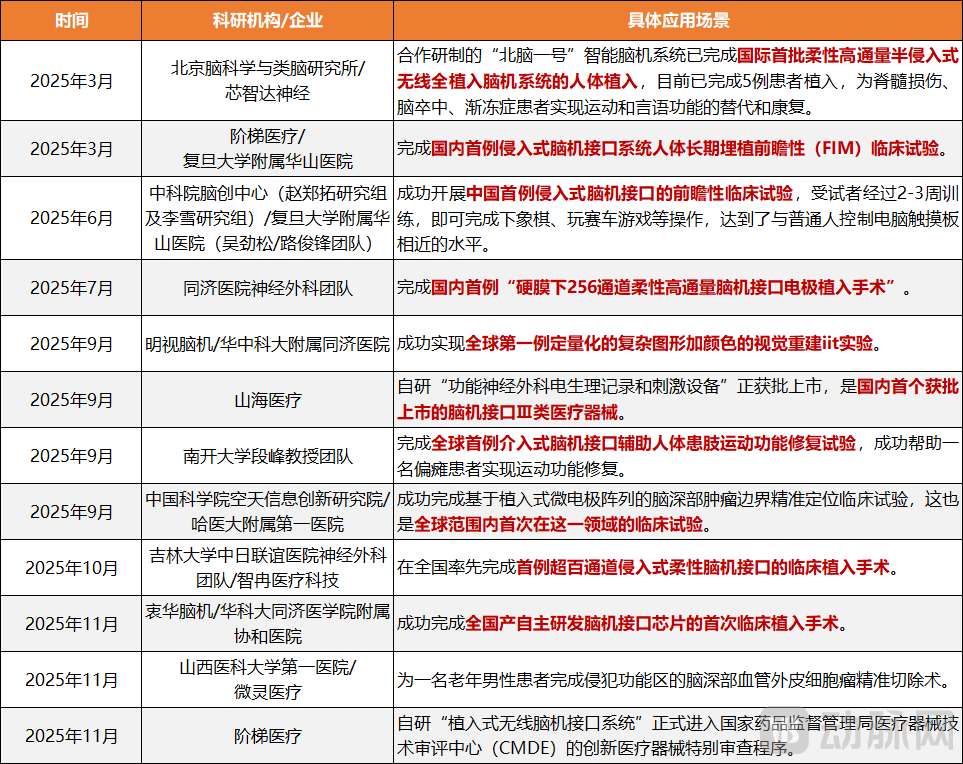
Figure 1. Breakthrough Clinical Research in China's Brain-Computer Interface Field by 2025
In the area of motor function restoration for patients with high-level paralysis, a team led by Professor Duan Feng from Nankai University conducted the world's first clinical trial of motor function rehabilitation in a paralyzed limb assisted by an interventional brain-computer interface in June 2025. By integrating interventional BCI technology with functional electrical stimulation, they successfully helped a hemiplegic patient achieve recovery of motor function in the affected limb. Reports indicate that following the implant procedure, the patient regained the ability to perform daily activities such as grasping freely and picking up medication with the left upper limb, demonstrating substantial overall improvement in motor function.
Regarding the restoration of speech and communication capabilities, early in 2025, Neuroxess, in collaboration with the Department of Neurosurgery at Huashan Hospital, Fudan University, and the Tianqiao and Chrissy Chen Institute (TCCI), successfully conducted clinical research on high-precision real-time motor and speech decoding. In this trial, a patient with brain injury mentally composed the phrase "Happy New Year 2025," which was decoded by a computer, ultimately sending a command to a robotic arm to form a heart-shaped gesture. It is noteworthy that decoding Chinese presents a greater challenge than English, which has only 26 letters, due to Chinese's 418 syllables and 4 tones. This complexity necessitates neural encoding and decoding mechanisms, along with information processing methods, specifically tailored to the characteristics of the Chinese language.
Finally, in the alleviation and treatment of mental disorders, in November 2025, a team led by Dr. Sun Bomin at Ruijin Hospital published data from China's first prospective clinical trials of invasive brain-computer interfaces for treatment-resistant depression in a Nature subsidiary journal. During the open-label phase, half of the patients (13 out of 26) experienced significant improvement in depressive and anxiety symptoms, with 9 of these patients achieving a state of remission where symptoms were essentially absent.
Commenting on these developments, Liu Bing, Founder of Mindtrix Technology, summarized, "Currently, the clinical applications of invasive brain-computer interfaces primarily focus on two major directions. One is neuromodulation, for conditions such as epilepsy or Parkinson's disease. The other is the reconstruction of neurological functions, including motor, visual, and language capabilities. In contrast, non-invasive BCI, leveraging its safety and ease of use, currently focuses more on consumer-grade interactive scenarios such as mood monitoring, attention training, health metric assessment, and simple command recognition."
How to Survive if It cannot Be Monetized on a Large Scale in the Short Term?
On November 11, 2025, the "Implantable Wireless Brain-Computer Interface System," independently developed by StairMed Technology, officially entered the special review process for innovative medical devices at the Center for Medical Device Evaluation (CMDE) of the National Medical Products Administration (NMPA). This marks China's first invasive BCI product to enter this "green channel," representing a critical step in the industrialization of brain-computer interfaces in China, transitioning from clinical validation toward market access.
While this progress is encouraging, it is essential to recognize that achieving large-scale commercialization in the BCI field will likely require at least 3-5 more years. Consequently, a pressing challenge now confronts all BCI companies: in the absence of stable commercialization and mature profit models, how can they survive in the interim?
To address this question, VCBeat has identified the following three viable pathways through case studies and interviews with industry experts:
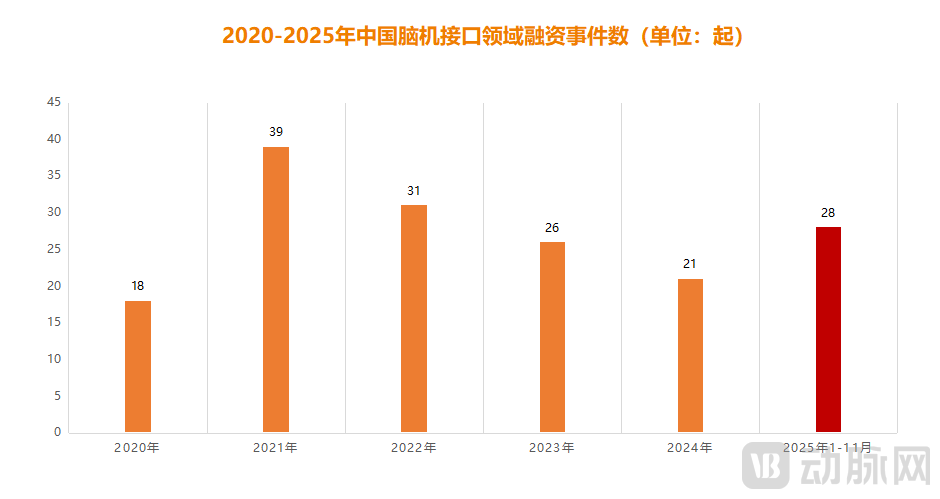 Figure 2. Number of BCI Financing Events in China (2020-2025). Data Source: VBData
Figure 2. Number of BCI Financing Events in China (2020-2025). Data Source: VBData
First, securing investment and financing serves as the essential lifeline. According to incomplete statistics from the VBData, the Chinese brain-computer interface (BCI) sector has witnessed over a hundred financing rounds in the past five years. These funds have been critical in sustaining many companies to date. Looking forward, raising capital remains the primary—and often the only—channel for financial infusion for many BCI firms. Therefore, survival hinges on possessing the core capability to secure continuous financing in the capital markets.
As the industry evolves, the underlying logic of BCI investment has shifted significantly. Jiang Donghui, Partner at Delian Capital, noted, "In earlier years, China's technological development path in BCI was unclear, and investments were primarily allocated based on upstream technological advantages. Recently, with increased engineering maturity and deeper clinical understanding of BCI in China, investors now evaluate potential targets not only on their technology's advancement, maturity, and feasibility in addressing real clinical needs but also on the core team's depth and breadth of scientific insight, ability to mobilize interdisciplinary resources, and drive for commercialization. In short, both the 'venture' and the 'team' must align, with a greater emphasis on market certainty."
This evolving logic inherently demands that BCI companies define a clear commercial pathway, which constitutes the second strategy for survival: focusing on a well-defined and viable track. Liu Bing, Founder of Mindtrix Technology, stated, "We should not always pursue overly broad ambitions or persistently aim to be the first to obtain regulatory approval in a specific domain. It is more crucial to first identify a promising track—one that addresses a rigid clinical demand, has a clear payment rationale, and is feasible to develop with currently available technology."
The third pathway involves generating interim revenue to achieve self-sufficiency and sustain operations. To this end, Jiang Donghui, Partner at Delian Capital, proposed a short-, medium-, and long-term strategy: "China has significantly increased investment in brain science research in recent years. Many innovative products from BCI companies can be applied and commercialized in research settings without regulatory approval, making the research market a short-term opportunity. In the medium term, companies can focus on upgrading existing clinical products, such as flexible stereoelectroencephalography (SEEG) electrodes, intraoperative monitoring devices, and innovative integrations with existing neuromodulation products. The long-term goal remains the development of BCI products targeting high-need clinical applications, advancing through clinical trials, and achieving regulatory approval and commercialization."
Overall, the key to survival for BCI companies today lies in securing greater market certainty—through the ability to sustain financing, a concentrated focus on a promising track, and the establishment of a viable commercialization path.
Rapidly Growing Market and Urgent Problems to Be Solved
According to data from Precedence Research, the global brain-computer interface (BCI) market was valued at approximately $2.62 billion in 2024 and is projected to reach $12.4 billion by 2034, reflecting a compound annual growth rate (CAGR) of 17.4% from 2025 to 2034. Focusing on the Chinese market, projections indicate that China's BCI market size will exceed 6 billion by 2028, with an expected CAGR of 17.7% from 2024 to 2028.
This remains a blue ocean market full of potential, yet the industry faces several distinct challenges. The first lies in technological bottlenecks, including insufficient accuracy of core algorithms and the immaturity of implantable materials and surgical techniques. These limitations can significantly compromise clinical performance and may even raise safety concerns.
The second challenge is a shortage of specialized talent. While China boasts considerable expertise in key areas such as electrodes and chips, there is still a scarcity of interdisciplinary professionals dedicated specifically to BCI. This talent gap makes it difficult for many companies to adopt a comprehensive perspective in systematically bridging technological R&D with clinical application, thereby hindering their normal development.
Finally, market education presents a significant hurdle, manifesting in two key aspects. On one hand, a lack of public trust in BCI technology persists, with considerable skepticism regarding its safety. On the other hand, ethical concerns are prominent. Many people worry that implanted BCIs, which continuously collect and decode vast amounts of neural signals, could lead to breaches of data privacy and security. The fear is that intruders might access "the brain's whispers," or that algorithms could potentially be used to manipulate human emotions with relative precision—a prospect that clearly conflicts with prevailing ethical norms and legal frameworks.
Fortunately, these issues are being progressively addressed. On the technological front, innovative technologies like Artificial Intelligence (AI) are being increasingly integrated into BCI applications. For instance, in signal processing and computation, AI serves as a powerful tool for signal extraction, compression, and calculation, enabling accurate interpretation of patient intent. Furthermore, regarding adaptability, AI allows BCI devices to engage in continuous learning and calibration, forming a closed-loop system based on execution feedback, which continuously enhances product performance.
Beyond technological advancements, a healthy industry ecosystem is rapidly taking shape. BCI is inherently a long-cycle, high-investment frontier field. Its sustainable advancement necessitates collaborative synergy across the entire chain—encompassing R&D, clinical practice, regulatory bodies, and capital investment. This requires forming deeply integrated partnerships aimed at co-innovation and shared value creation, while also jointly assuming risks. The collective goal is to accelerate the transition of BCI technology from "laboratory breakthroughs" to "clinical adoption," ultimately achieving industrial scale.
Nicolelis, often regarded as a founding father of BCI, once stated that the true development and widespread adoption of BCI technology must meet three primary standards: safety, efficacy, and affordability. Therefore, as China's BCI sector pivots critically towards clinical translation, these principles are becoming the gold standard, effectively selecting for the "long-distance runners"—those enterprises genuinely capable of delivering medical-grade solutions and successfully navigating the complexities of regulatory approval and reimbursement.
