From world-first intrathecal injection to 10 INDs: NovaBio accelerates clinical translation of China-developed Treg therapy
Cell and Gene Therapy (CGT) is transitioning from immune activation to a new phase of immune homeostasis reconstruction.
If the first wave of innovation, represented by CAR-T therapies, drove breakthroughs in cancer treatment through immune activation, the current focus of global research and industry is gradually shifting towards the reconstruction and restoration of homeostasis in the immune system. In this shift, Regulatory T cells (Tregs) are recognized as one of the most promising research directions. The awarding of this year's Nobel Prize in Physiology or Medicine for research related to Treg mechanisms has further intensified global attention on this field.
Tregs play a crucial role in maintaining immune balance by regulating immune responses and suppressing excessive inflammation. Currently, no Treg cell therapy has received market approval globally, but numerous early-stage clinical trials are underway across various disease areas. Existing research indicates the potential therapeutic value of Tregs in neurodegenerative, autoimmune, and fibrosis-related diseases, all areas characterized by significant long-term unmet medical needs.
"The immune system is not simply about attack and defense; it is a dynamic system that requires precise regulation," said Dr. Lu Mingqi, Founder and CEO of NovaBio, in an interview. "Our core focus is not merely suppressing immunity, but restoring balance—enabling the immune system to operate again within an appropriate range."
Based on this philosophy, NovaBio has chosen to start with Treg therapies, taking the lead in China in exploring their path from scientific research to industrial application.
The Foundation for Treg Clinical Translation
The founding of NovaBio originates from the accumulation and judgment of its founder, Dr. Lu Mingqi, gained through over thirty years in the global drug R&D field. Under the mentorship of Peter Agre, the 2003 Nobel Prize Laureate in Chemistry, Dr. Lu previously led the development of multiple categories of innovative drugs—including Programmed Death-1 (PD-1) antibodies and Antibody-Drug Conjugates (ADCs)—at several multinational pharmaceutical companies, thereby fully experiencing the technological evolution from small molecules and antibodies to cell therapies.

Figure 1: Dr. Lu Mingqi, Founder and CEO of NOVABIO
Years of mechanistic research led him to recognize that many complex diseases do not stem from "immune deficiency" but from dysregulation of immune control. Traditional drugs often only alleviate symptoms and struggle to restore balance at a systemic level. "Tregs are the cell type capable of systemically restoring immune homeostasis. They are not killer cells; they calm the immune system down," he summarized in the interview.
Based on this understanding of disease mechanisms, Dr. Lu Mingqi founded NovaBio in 2022, with formal operations launching in 2023. The company established a Treg cell therapy-focused technological strategy, adhering to a direction guided by unmet medical needs. Rather than chasing short-term trends, NovaBio is building systematic capabilities in the still-nascent Treg field. "Speed must be built upon quality; systematic capability is the key to improving efficiency," Dr. Lu emphasized. Currently, NovaBio already has projects entering the Investigator-Initiated Trial (IIT) stage and has submitted multiple IND applications, achieving zero deficiency feedback in its first IND and orphan drug submissions to both Chinese and US regulatory agencies.
NovaBio's development pace benefits significantly from the team's structured division of labor and accumulated experience. Key functions—including R&D, quality control, process scale-up, and regulatory affairs—are staffed by members with backgrounds in both Chinese and international pharmaceutical companies and research institutes. Several core team members possess experience in IND design and CMC from multinational pharma, enabling NovaBio to establish, at its early corporate stage, integrated processes covering from process development to quality systems.
Capital and clinical networks have also accelerated the company's growth. In April 2023, NovaBio secured approximately 44 million in Angel round funding. In 2024, its core product NP001 (Autologous Polyclonal Treg Cell Injection) received FDA Orphan Drug Designation and completed the world's first intrathecal injection in an ALS (Amyotrophic Lateral Sclerosis) patient. In 2025, the company is advancing a Pre-A+ round, planning to raise 100–200 million to support pivotal clinical trials and pipeline expansion.
Starting from disease mechanisms, building a foundational system through process breakthroughs, and progressively strengthening clinical networks and capital support, NovaBio is constructing a comprehensive Treg industrialization path spanning from research to clinical translation.
Multi-Dimensional Treg Pipeline Matrix
If "vision and speed" were the initial hallmarks that made NovaBio recognizable within the industry, what has truly enabled it to advance multiple clinical programs in a short time and secure a firm foothold in the global Treg landscape is a foundational technology system built upon quality, scalability, and scientific rigor.
In the field of cell therapy, Tregs have long been considered one of the most challenging directions: they constitute only 5–7% of peripheral blood, presenting longstanding industry-wide challenges such as difficult isolation, difficult expansion, susceptibility to differentiation, and functional attenuation. To address these bottlenecks, NovaBio has developed its own high-purity Treg manufacturing platform, systematically optimizing the entire process—from cell separation reagents and culture systems to quality control systems. This enables stable expansion of Tregs by several hundred-fold, controls impurity cell rates below 1%, and ensures continuous scaling capability from research-grade to GMP-grade. As Dr. Lu Mingqi stated, "Every batch of cells is tested for inhibitory function according to international standards and is only released after confirming its ability to effectively control aggressive immune cells. Our quality standards are no lower than those of any institution worldwide."
Building on this manufacturing foundation, the company has further established six R&D platforms and secured multiple global patents: Polyclonal Tregs, Targeted Tregs, Universal Tregs, Treg-derived Exosomes, In Vivo-Induced Tregs, and a Small Nucleic Acid Drug Platform. These cover multiple pathways, from cell-based entities to exosomes, and from in vitro expansion to in vivo induction. Such a matrix-based layout provides NovaBio with greater extensibility in process development, delivery systems, indication selection, and product forms.
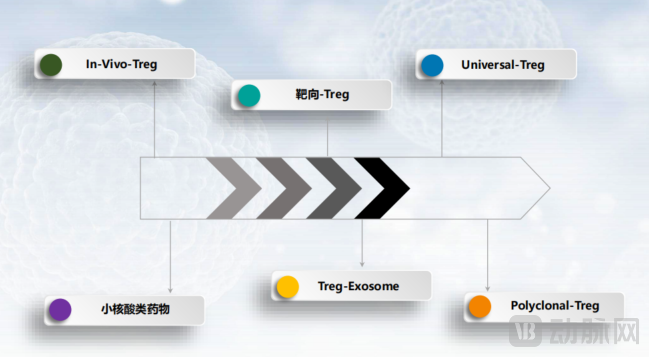
Figure 2: NOVABIO Treg Product Platform Layout
The breakthrough in core technology lies in its innovative route of administration. Conventional intravenous injection of Tregs struggles to cross the blood-brain barrier (BBB), limiting their potential for central nervous system (CNS) diseases. NovaBio employs intrathecal injection to deliver Tregs directly into the cerebrospinal fluid (CSF) circulation, thereby bypassing the BBB and reaching the lesion site. "We were the first team globally to deliver Tregs directly into the CSF circulation," recalled Dr. Lu.
In animal studies using SOD1-mutant mouse models of ALS, promising efficacy results were achieved, showing significant delays in disease onset and extension of survival. This approach has also demonstrated initial value in Investigator-Initiated Trials (IITs): some patients transitioned from declining ALSFRS-R scores to stabilization or even mild improvement, with no serious adverse events observed.
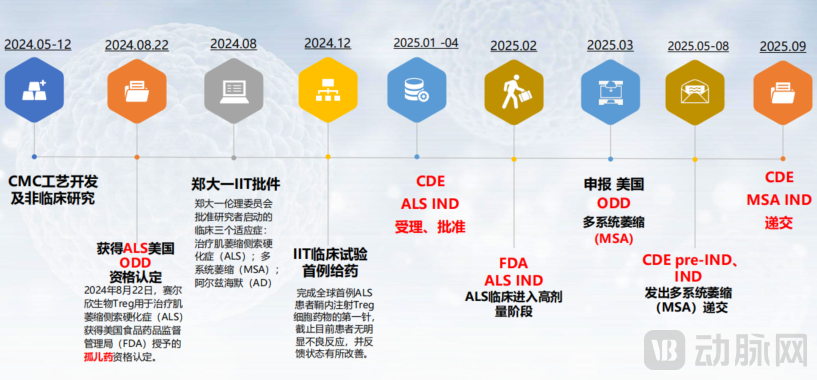
Figure 3: NP001 Key Milestone Events
From commencing operations in 2023 to achieving the world's first intrathecal delivery of Tregs in 2024, NovaBio accomplished this milestone in just over a year. The company plans to submit 10 IND applications between 2025 and 2026. This rapid progress is not achieved by merely allocating more resources, but is a natural outcome of synergies among stable manufacturing, innovative delivery technologies, and a well-integrated platform system.
The Chinese Path to Treg Industrialization
Globally, the Treg cell therapy field remains largely in early exploratory stages, with most companies still in preclinical research or focusing on major diseases with established research foundations. For instance, Sonoma Bio, co-founded by Nobel Laureate Fred Ramsdell, has raised over $360 million USD, yet its core CAR-Treg program SBT-77-7101—currently in Phase I clinical trials for rheumatoid arthritis—is still in early development. Similarly, Coya Therapeutics' polyclonal Treg therapy COYA 101 is also in early clinical stages, with no public documentation of IND approvals for either company.
Against this industry backdrop, NovaBio has chosen to avoid homogeneous competition by concentrating on truly unmet medical needs. It has built a pipeline targeting three major therapeutic areas—neurodegenerative diseases, autoimmune diseases, and metabolic diseases—through innovations in delivery routes and platform systems. Adopting a "rare diseases first, common diseases later" strategy, the company aims to validate mechanisms in high-unmet-need areas before expanding into broader indications. Its current pipeline includes six core programs, utilizing differentiated routes of administration such as intrathecal and intravenous injections tailored to specific indications, making NovaBio the first company in China to advance Treg cell therapies into registered clinical trials.
Compared to the progress of global peers, these advancements not only demonstrate NovaBio's leadership in clinical translation speed and systematic execution but also provide a referable technological and industrial model for immune homeostasis therapies in China. As Dr. Lu Mingqi emphasized, "Our work on Tregs is not for the sake of innovation itself, but to address genuine unmet medical needs. Compared to conditions like type 2 diabetes, which already have symptomatic treatments, rare diseases such as ALS and MSA (multiple system atrophy) remain without effective therapies. We are committed to tackling these tough challenges."
To enhance clinical research efficiency and support global deployment, NovaBio is establishing collaborative networks with leading medical institutions in China, including Beijing Tiantan Hospital, Peking Union Medical College Hospital, and The First Affiliated Hospital of Zhengzhou University, forming a multi-center research network focused on neurodegenerative and autoimmune diseases. The company is also partnering with Haier Biomedical to build an international cell bank and overseas clinical platform in preparation for future multi-regional trials. Additionally, collaborations with Union Hospital, Tongji Medical College of Huazhong University of Science and Technology are advancing research on rare diseases such as Kennedy's disease and Huntington's disease. These partnerships reflect a systematic approach characterized by clear objectives, complementary capabilities, and comprehensive coverage in the Treg field.
While continuously advancing its technology, NovaBio is also promoting the local production of key technologies in China to enhance manufacturing stability and accessibility. Dr. Lü noted in the interview, "If key processes can be established within China, it will not only improve stability but also make therapies more accessible to patients." This synergistic combination of a proprietary technology system, localized production, and multi-center clinical capacity is forming a distinctive pathway in the cell therapy industry.
Looking ahead three to five years, NovaBio plans to advance one or two Treg products toward marketing approval and initiate global multi-center studies. "We hope to enable real and effective treatments for ALS and MSA patients as soon as possible, and we encourage more companies to pursue innovation driven by medical needs," Dr. Lu stated. This vision reflects a broader trend of Chinese biopharmaceutical companies evolving from followers to definers in the global landscape.
From focusing on rare diseases to expanding into common conditions, and from cell processing to clinical systems, NovaBio presents an exploratory model for immune homeostasis therapies from China. In Dr. Lu Mingqi's view, "solving clinical problems" remains the company's core logic. Accelerating the delivery of scientific achievements to patients represents the ultimate significance of Tregs' breakthrough in China.
