Oral height-growth pills: not a scam anymore?
"All oral height-growth pills are scams, nothing more than an 'IQ tax'!"
For years, these so-called "oral height-growth pills" have been riding the coattails of legitimate prescription growth hormone injections. Reliable health information online has consistently warned parents about these fraudulent products. But this widely held understanding may be about to change.
Recently, GeneScience Pharmaceuticals has received clinical trial approval for GS3-007a - an oral small-molecule growth hormone secretagogue in dry suspension form. Taken once daily, GS3-007a stimulates the body's natural growth hormone production, potentially offering a new treatment option for children with diagnosed short stature.
This development suggests that scientifically-validated oral height-growth pills could become a reality. It also signals that the future competitive landscape of the growth hormone market will likely evolve - shifting from the rivalry between long-acting and short-acting formulations to a new competitive front: oral versus injectable delivery.
Since recombinant human growth hormone (rhGH) was first approved in the 1980s, daily subcutaneous short-acting formulations have long dominated the market. However, this treatment regimen has limitations: frequent injections not only cause distress to pediatric patients but also lead to widespread missed doses or premature treatment discontinuation, ultimately compromising therapeutic outcomes.
According to data from Frost & Sullivan, a 2021 study revealed that 82% of pediatric growth hormone deficiency (PGHD) patients missed at least one of their daily injections per week. In another study involving 110 children, two-thirds of patients missed, on average, more than one injection per week. Patients who missed injections more than once per week showed a clinically relevant reduction in the change of height velocity standard deviation score compared to those who missed injections no more than once per week (where a higher score indicates faster growth).
To address these challenges, long-acting growth hormones have become a key focus of current R&D, primarily featuring once-weekly dosing. Using technologies such as PEGylation, Fc fusion, and albumin binding, these long-acting formulations extend the drug's half-life in the body, reduce dosing frequency, and thereby improve patient adherence.
To date, China has approved two long-acting growth hormone products: GeneScience's Jintropin AQ Polyethylene Glycol Recombinant Human Somatropin Injection and Amoytop Biotech's Pegpesen® Inpegsomatropin Injection.
Among them, Jintropin AQ, launched in 2014, has accumulated extensive clinical experience. According to the financial report of GeneScience's parent company, Changchun High-tech, a 5-year real-world study based on the China Growth Hormone Long-term Efficacy and Safety Assessment (CGLS) database, published in June 2025, demonstrated long-term safety in 1,207 cases continuously treated with PEG-rhGH for up to five years. The study reported no unexpected adverse reactions and no drug-related serious adverse events.
Pegpesen®, approved in 2025 as a Category 1 new drug, is a new-generation long-acting growth hormone modified with 40 kDa Y-shaped branched polyethylene glycol in a single-molecule configuration. By optimizing the modification strategy—primarily targeting non-N-terminal sites—the product achieves higher specific bioactivity and an extended half-life. This allows for maintained efficacy while reducing the required dose, thereby offering improved long-term drug safety.
In addition, the Biologics License Application (BLA) for Visen Pharmaceuticals' Lonapegsomatropin for Injection has been accepted and is expected to gain approval in the fourth quarter of 2025.
Lonapegsomatropin is a long-acting growth hormone that Visen Pharmaceuticals licensed from Ascendis Pharma in 2018. It previously received marketing approval from the U.S. FDA in August 2021 and the European Medicines Agency (EMA) in January 2022. Public data show that the overseas sales of Lonapegsomatropin reached €179 million in 2023 and €202 million in 2024, approximately RMB 1.502 billion and RMB 1.695 billion, respectively, demonstrating steady growth.
Meanwhile, several other long-acting growth hormone candidates remain under development in China, most of which have already entered the clinical trial stage. Typically, it takes approximately 1 to 5 years to advance a drug from Phase III or Phase II clinical trials to market approval. Therefore, within the next five years, China is expected to approve an additional 4 to 5 long-acting growth hormone products.
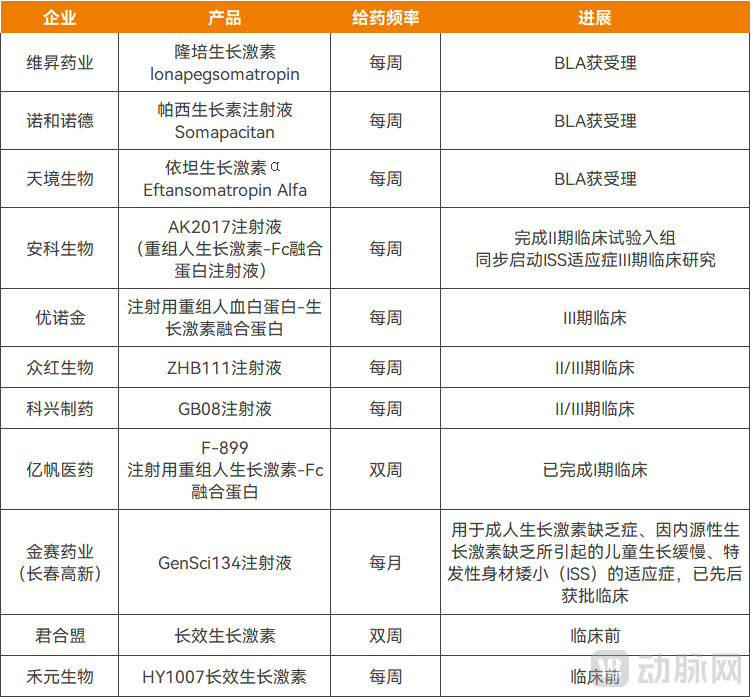
Figure1. Long-acting growth hormones currently in the research and development stage in China.
Source: Corporate financial reports, prospectuses, and other public information
Regarding further optimization of dosing frequency, a trend toward biweekly and even monthly administration is emerging.
For example, JHM Biopharmaceutical's investigational long-acting growth hormone JunoRegen™ utilizes genetically engineered E. coli technology with an Fc fragment as a long-acting carrier. Through inclusion body renaturation, assembly, and multi-step purification processes, the drug's half-life is significantly extended. Preclinical studies indicate its potential for biweekly dosing.
GeneScience's investigational GenSci134 injection exerts pharmacodynamic effects through specific targeting of the growth hormone–insulin-like growth factor-1 (IGF-1) pathway. Preclinical data demonstrate that GenSci134 features a unique molecular design and metabolic profile. Animal PK/PD modeling based on non-clinical data suggests its potential for monthly dosing.
In summary, although short-acting growth hormones currently dominate the market, the launch of more long-acting formulations is expected to accelerate the transition toward a long-acting and even ultra-long-acting era.
While long-acting injectables address the issue of dosing frequency, they still present unavoidable challenges: some children exhibit strong aversion to injections; long-term subcutaneous administration may lead to adverse reactions such as local lipoatrophy and induration; and the injection process requires a certain level of expertise, creating inconvenience for both children and their parents.
Against this backdrop, oral medications have emerged as a longer-term goal for the industry. GeneScience's GS3-007a dry suspension represents a significant exploration in this direction.
As an oral small-molecule growth hormone secretagogue, GS3-007a is intended for the treatment of growth retardation in children caused by endogenous growth hormone deficiency. With once-daily oral dosing, it stimulates the release of endogenous growth hormone, potentially offering additional treatment options for children with short stature who require growth promotion.
The development of oral formulations for growth hormone faces significant technical hurdles, chief among them being the protein's nature. After oral administration, it is susceptible to degradation by stomach acid and proteases in the gastrointestinal tract, resulting in low bioavailability. Additionally, the large molecular size of proteins makes it difficult for them to cross intestinal epithelial cell membranes for absorption.
GS3-007a adopts a small-molecule secretagogue approach: rather than directly supplying exogenous growth hormone, it stimulates the pituitary gland to secrete endogenous growth hormone via an oral small-molecule drug, thereby circumventing the problem of protein degradation.
Globally, Lumos Pharma's LUM-201 also adopts a similar technical approach. As a growth hormone secretagogue receptor (GHSR) agonist, LUM-201 met both primary and secondary endpoints in two Phase II clinical studies, demonstrating therapeutic efficacy for pediatric growth hormone deficiency (PGHD).
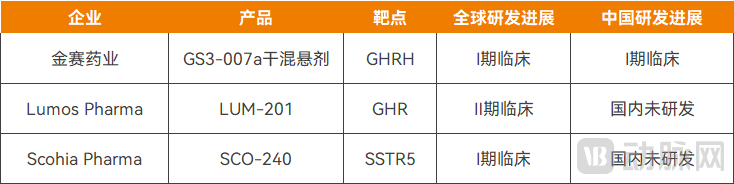
Figure 2. Oral Growth Hormone Drugs Under Research Worldwide.
Source: Pharnexcloud Database, Public Information from Companies
Additionally, Scohia Pharma's SCO-240 is designed as an oral selective somatostatin receptor subtype 5 (SSTR5) antagonist. As a small molecule, it stimulates growth hormone secretion without affecting other pituitary hormones, with clinical data demonstrating favorable safety and tolerability.
According to public data compiled by VCBeat, only the three aforementioned oral drugs for growth hormone deficiency are currently in clinical trial stages worldwide.
Undoubtedly, oral medications will bring disruptive change to growth hormone administration methods. However, the current investigational drugs remain in relatively early clinical stages, and substantial time is still required before commercialization.
It is worth emphasizing that while dosing convenience represents one of the most anticipated advantages of oral drugs, realizing this benefit requires demonstrating non-inferior efficacy and reliable safety compared to short-acting growth hormone treatments, while also accounting for multiple practical factors in the transition from product to commercially viable therapy.
According to data from the Pharnexcloud Database, there have been nine oral drug candidates globally for growth hormone deficiency. As of now, only the three pipelines mentioned earlier remain active in development, while the others are either in a "no subsequent progress reported" status or have had their studies discontinued.
Among these, Rani Therapeutics' RT-109 is one such discontinued pipeline. RT-109 attempted to use the company's self-developed capsule robot, RaniPill, for drug delivery. The capsule is designed to release microneedles after entering the intestine, thereby injecting growth hormone into the submucosal layer of the intestinal wall. RaniPill was also intended to replace injectable administration of other biologics with an oral approach.
Changchun High-tech (GeneScience's parent company) also previously entered into a collaboration with Rani Therapeutics, securing an evaluation and option agreement for RT-109, which included priority negotiation rights for its commercialization in China. However, by 2022, Rani Therapeutics announced the discontinuation of RT-109 research, primarily due to issues related to the shipment of raw material samples.
In summary, the successful launch of oral drugs still requires overcoming multiple significant challenges. Although the path forward is long and uncertain, the three current investigational candidates have demonstrated efficacy and safety in preliminary clinical trials, indicating a reasonable probability of success, and their future market potential is worthy of attention.
In the future growth hormone market, whether for injectables or oral drugs, biologics or chemical drugs, and regardless of the technological approach adopted, one unavoidable reality must be faced: the size of the potential patient pool is contracting. A review of financial reports from major listed companies in the growth hormone sector reveals an overall downward trend in sales revenue or profits over the past two years.
PGHD primarily affects children from newborns to 17 years old. Against the backdrop of declining birth rates, the number of PGHD patients is also projected to decrease from 2023 to 2030. However, as calculated by Visen Pharmaceuticals in its IPO prospectus, despite the reduction in patient numbers, the treatment rate for PGHD is expected to more than double between 2023 and 2030.
According to data from Frost & Sullivan, China's PGHD treatment rate is projected to increase from 5.3% in 2023 to 10.7% by 2030. The continuous improvement in diagnosis rates, coupled with the development of safer and more convenient treatment options, will directly drive the increase in treatment rates.
This suggests that, theoretically, the growth hormone market still has room for expansion. New entrants and newly launched products still have opportunities to secure a place in the market.
However, increasing the treatment rate requires the combined effect of multiple factors, such as: advances in diagnostic technology, particularly the continuous improvement of screening capabilities in primary healthcare institutions, leading to the identification of more potential patients; enhanced health awareness among parents, increased attention to children's height, and greater willingness to seek medical consultation proactively; and optimization of treatment options, including the availability of more convenient products such as long-acting formulations and oral medications, which lower the treatment barrier for patients.
Therefore, companies must prioritize these areas to drive increases in treatment rates. This includes expanding into primary care and out-of-hospital markets, developing new growth opportunities, and enhancing market awareness through healthcare provider and patient education. Furthermore, product-specific improvements in logistics and storage convenience are essential. For instance, while most growth hormone products historically required cold chain transportation and low-temperature storage, Visen Pharmaceuticals' lonapegsomatropin used with its auto-injector device enables home storage at room temperature for up to six months—enhancing the patient treatment experience and creating a competitive advantage.
In addition, indication expansion will serve as another key pathway for growth hormone products to enhance market penetration.
Pediatric Growth Hormone Deficiency (PGHD) is the most common form of short stature in patients under 18 and the most prevalent indication for growth hormone therapy. Additionally, growth hormone can be used for various pathological short stature conditions, including Idiopathic Short Stature (ISS), Small for Gestational Age (SGA), Turner Syndrome, and Noonan Syndrome. During the era of short-acting formulations, Jintropin® (developed by GeneScience) achieved approval for up to 12 indications.
Growth hormone deficiency, which can occur at any stage of life, is a developmental and metabolic disorder caused by factors such as pituitary dysplasia, craniocerebral injury, genetic mutations, hypothalamic dysfunction, or chronic systemic diseases. Among these, Adult Growth Hormone Deficiency (AGHD) often presents with multiple complications and carries significant health risks.
Given the current treatment landscape, AGHD patients require long-term or even lifelong replacement therapy, representing an area of unmet clinical need. The Expert Consensus on the Diagnosis and Treatment of Adult Growth Hormone Deficiency (2025 Edition) also indicates that drug therapies for AGHD include both short-acting and long-acting growth hormone formulations.
Therefore, AGHD has become an important direction for indication expansion in growth hormone therapy.
Currently, based on substantial efficacy and safety data accumulated from PGHD treatments, Jintropin AQ is conducting a Phase III clinical trial in China for AGHD. In June 2025, GeneScience's ultra-long-acting growth hormone—GenSci134 injection—received approval to initiate clinical trials for AGHD.
Amoytop Biotech's Pegpesen®, approved for PGHD at its initial launch in May 2025, also gained approval in August for a new clinical trial in AGHD.
In summary, the growth hormone market is at a critical juncture of technological iteration and market restructuring, with multiple trends driving industry upgrading. Long-acting and ultra-long-acting growth hormone products offer significant advantages in reducing dosing frequency and improving patient adherence. Oral formulations, as the ultimate goal, are expected to reshape the industry landscape once technical barriers and challenges in the product-to-commercialization transition are overcome, potentially ushering in a new "golden decade" for the sector.
Nevertheless, the future direction of the market will depend on whether new products and technologies can first ensure safety and efficacy, effectively address specific clinical pain points, avoid misuse, and remain focused on addressing genuine patient needs throughout development and promotion.
